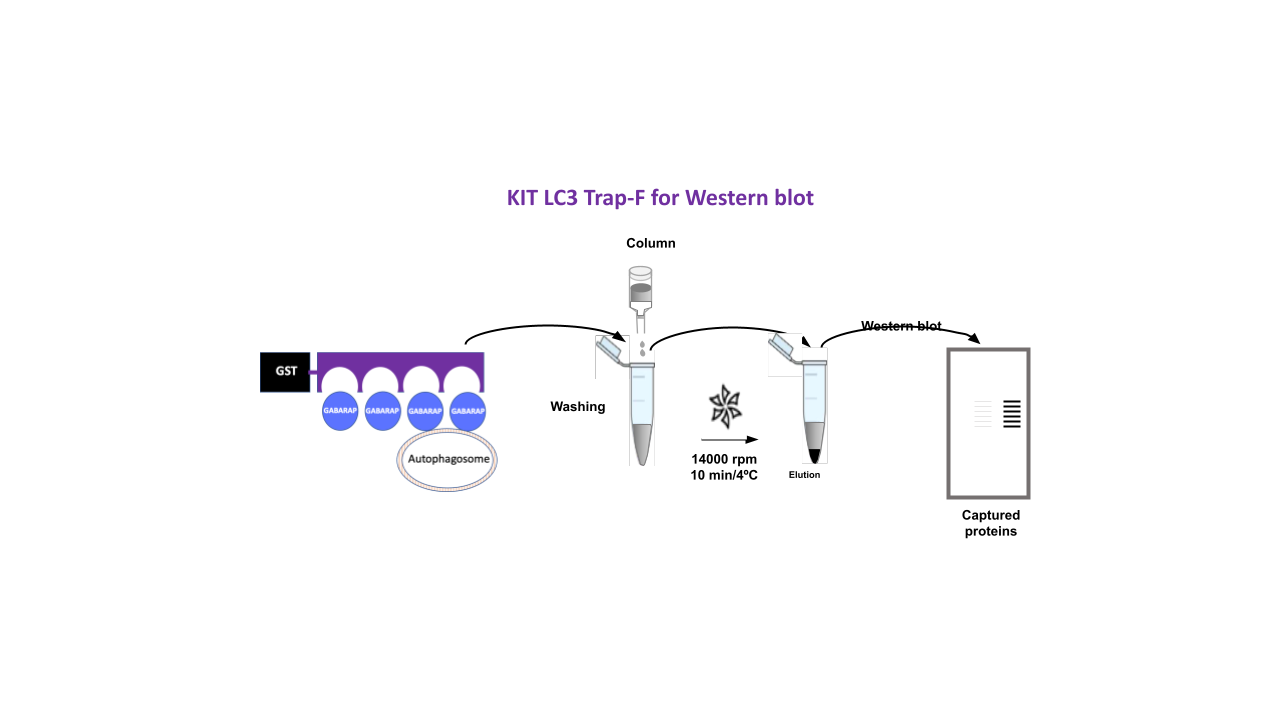
Applications
-Capture of GABARAP, GABARAP-L1 and GABARAP-L2 proteins as well as complexes associated to these proteins from cell lines and model organisms followed by Western blot detection.
-Immunoprecipitation of complexes associated to GABARAP, GABARAP-L1 and GABARAP-L2 proteins. This application requires the elution of captured proteins, dialysis and immunoprecipitation with a specific antibody recognizing a protein of interest that are not provided in this kit.
Specificities
LC3-traps F are not crosslinked to agarose beads but preserved in a lyophilized state for long term storage. LC3-traps F are reconstituted with a specific buffer just before use and mixed with glutathione agarose beads provided in the kit. Washing and elution buffers are also included. Negative control GST is provided for a single control reaction.
Captured proteins have to be boiled to be analyzed by Western blot. Since LC3-traps are not crosslinked to agarose, these will be released during the boiling. Avoid using antibodies for Western blot analysis that have been generated with a GST-protein of interest fusion, otherwise GST protein will be detected.
Characteristics
- Tag: GST
- Purity: > 95% by RP-HPLC and SDS-PAGE
- Molecular Weight: 41,54 kDa
- Physical State: Solid (powder)
- Quantity: 250 µg
- Solubility: <5 mg/mL
- Concentration: Variable
- Storage: RT but once reconstituted store at -80°C and avoid freeze/thaw cycles
Benefits
- Higher affinity than the single LIR domain
- Avoid overexpression of tagged ATG8 proteins for pull downs
- Replace LC3/GABARAP specific antibodies for enrichment of these proteins
- Kit LC3 Trap-F has been developed to facilitate the use, long-term stability and transport of all components required to capture proteins associated to LC3/GABARAP using agarose beads.
Examples of use
Kit LC3 Trap-F can be used to detect LC3/GABARAP proteins and cellular factors directly or indirectly associated to them implicated in the regulation of autophagy and signaling pathways regulating inflammation, oncogenesis, neurodegenerative diseases and multiple infections (3). The lipidated status of LC3/GABARAP proteins can be followed in response to a treatment and determine if cells are responding well (1, 2).
Publications
1. Quinet G, Génin P, Ozturk O, Belgareh-Touzé N, Courtot L, Legouis R, Weil R, Cohen MM, Rodriguez MS (2021) Exploring selective autophagy events in multiple biologic models using LC3-Interacting Regions (LIR)-based molecular traps. Scientific Reports. In press.
2. Quinet G, Ozturk O, Belgareh-Touzé N, Legouis R, Cohen MM, Rodriguez MS (2021) Capture of ATG8 family members using LC3-traps. MiMB. In press.
3. Jiang, P. & Mizushima, N. Autophagy and human diseases. Cell Res 24, 69–79 (2014).